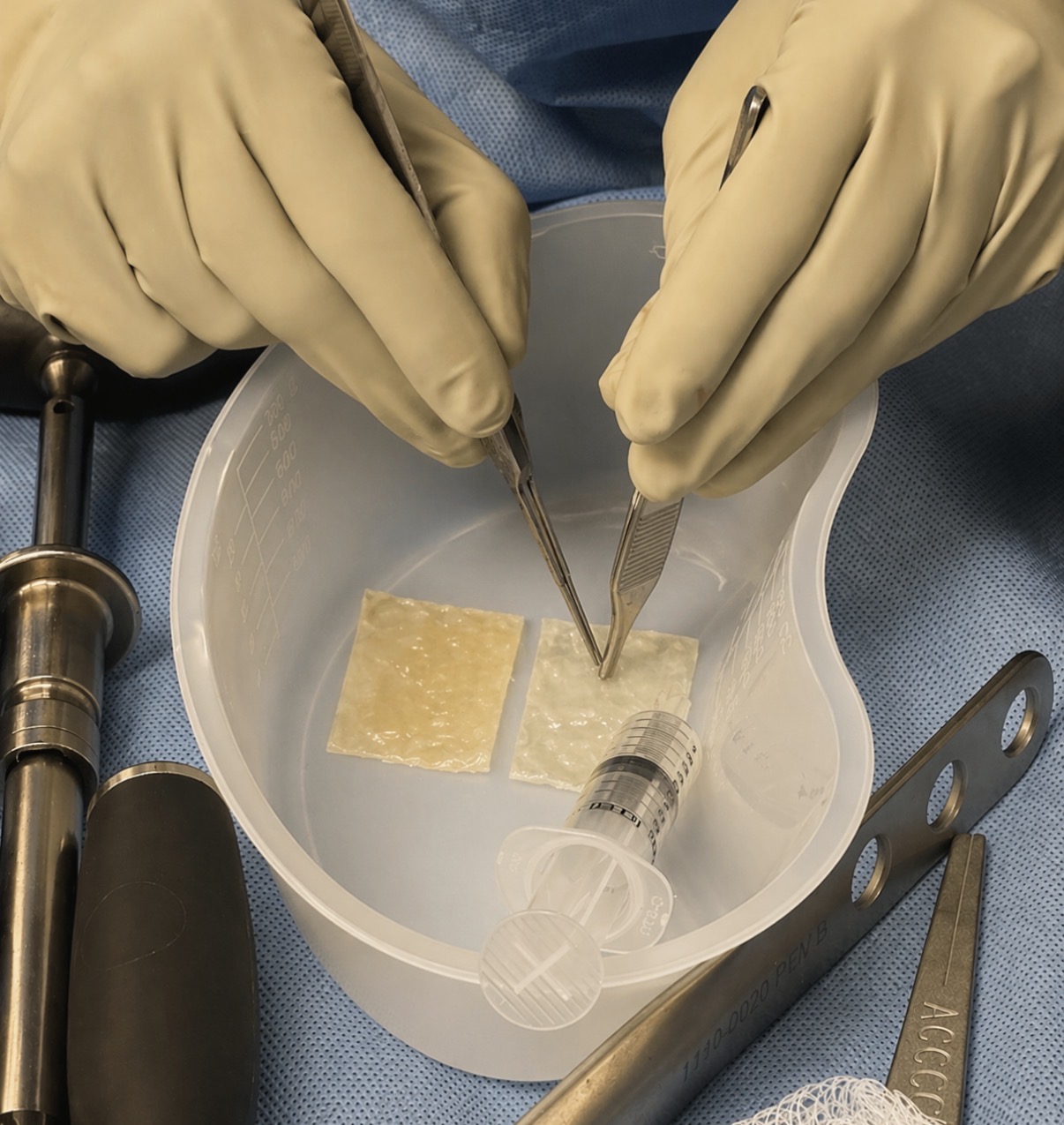
Structural Tissue Allograft (STA) is a full-thickness, hydrated placental matrix designed to supplement, repair, and reinforce native tissue. Unlike particulates or flowables, STA is a structural implant—preserving native architecture and providing a physical framework for intra-operative tissue support.
One implant. Multiple tissue environments.
Not a filler. Full-thickness. Ultra-minimally manipulated. Designed to provide a physical framework for surgical tissue support.
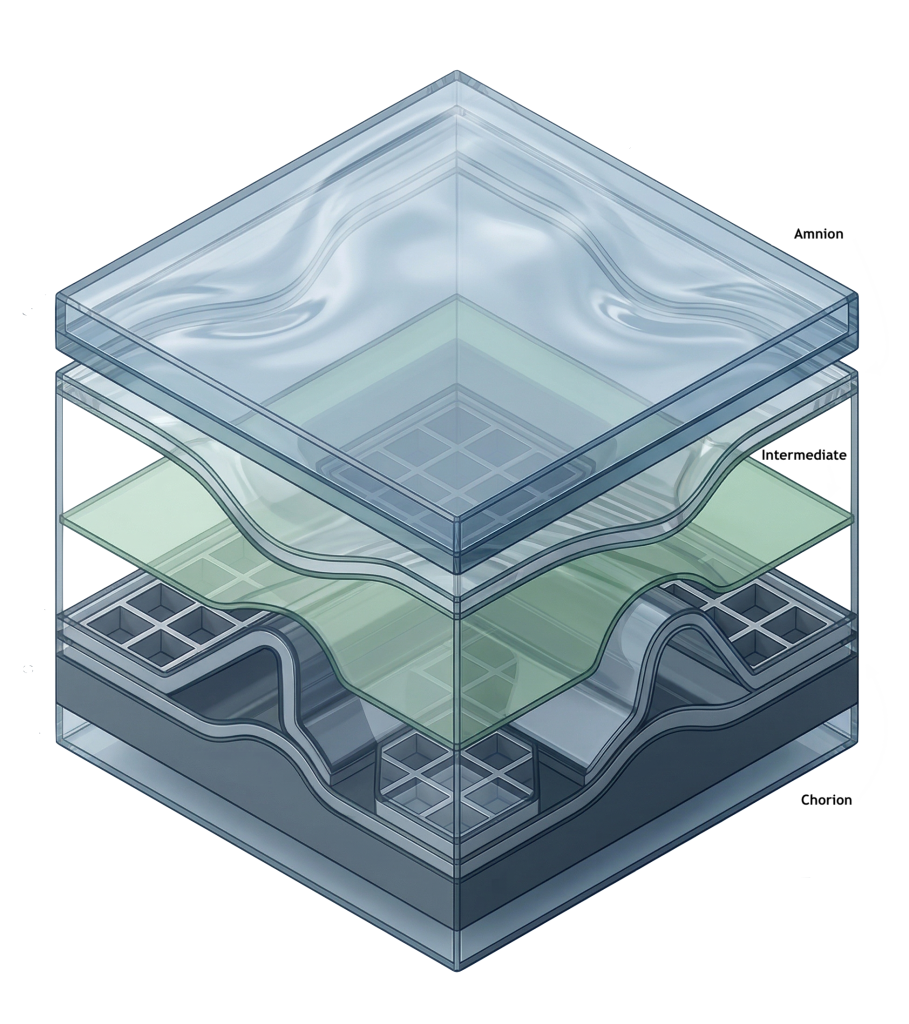
Preserves native tissue architecture—including collagen and elastin—maintaining structural continuity across layers.
361 HCT/P compliant structural scaffold that retains native extracellular matrix components, including fibronectin, laminin, and glycosaminoglycans.
Three structural components. One preserved system.
One implant. Multiple tissue environments.
Supports site-appropriate tissue repair across multiple environments.
Reinforces soft tissue structures.
Provides a structural interface for cartilege repair.
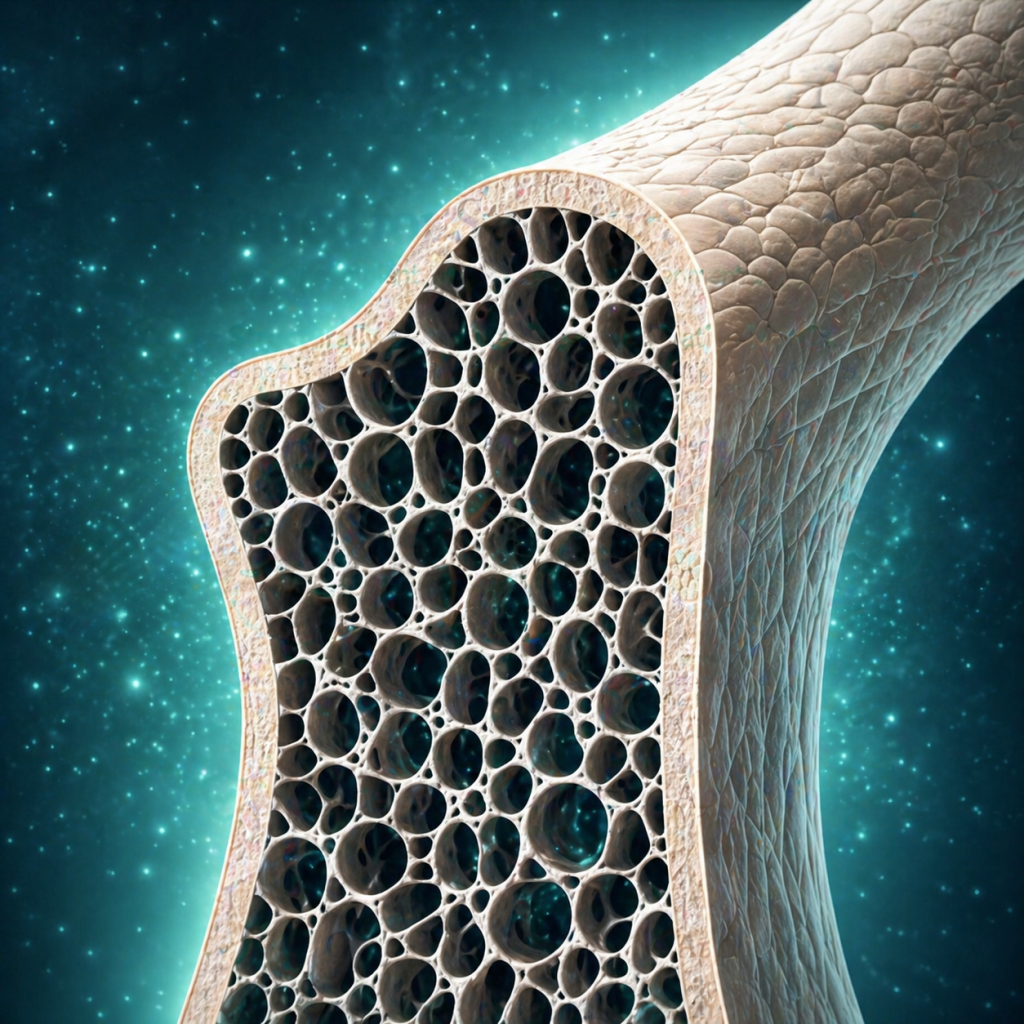
Supports structural integration in bone repair procedures.
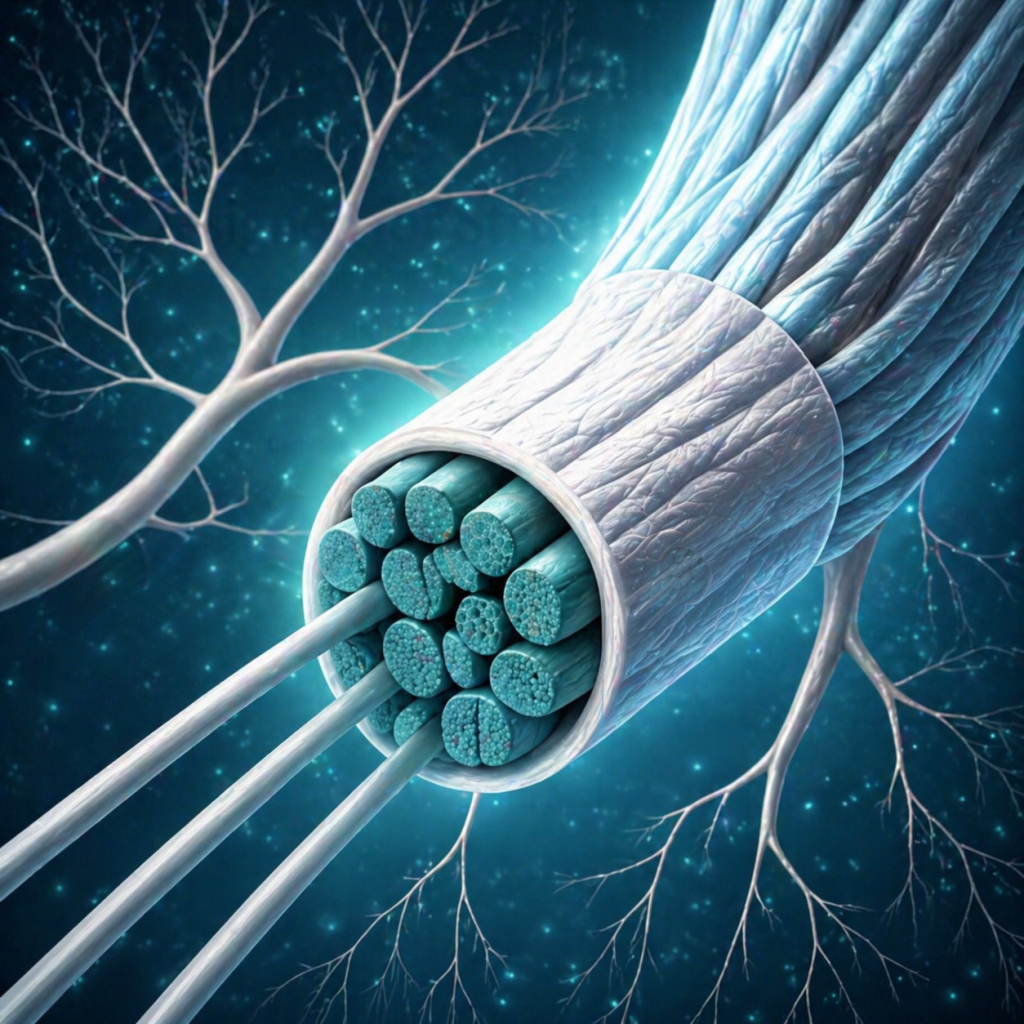
Provides a protective interface for delicate neural structures.
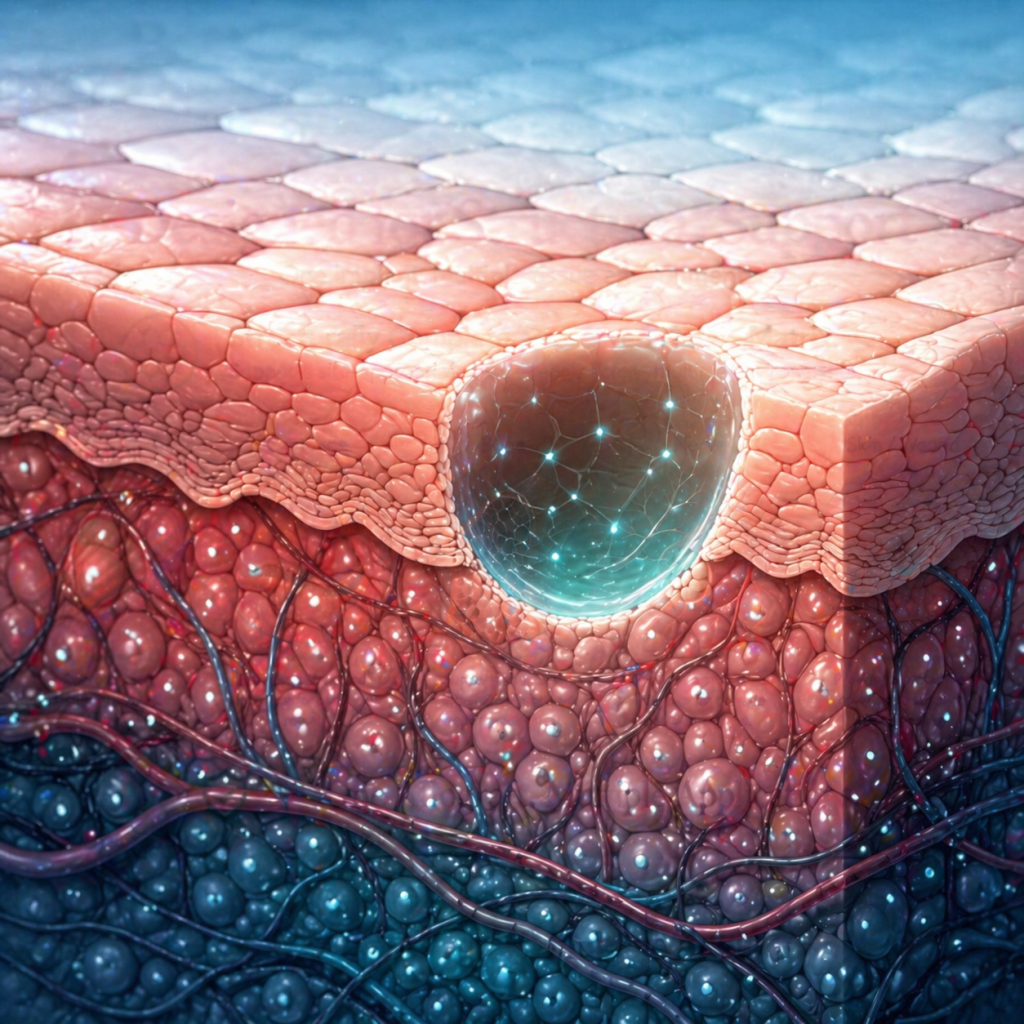
Provides a protective covering to support wound healing.
By preserving the native extracellular matrix in its hydrated state, STA maintains both structural integrity and the native composition of the tissue.This enables the implant to function as a functional structural scaffold, supporting organized tissue repair rather than acting as a passive patch.
Together, these structural components preserve the native tissue environment, supporting organized tissue remodeling.
Enables consistent product utilization across Orthopedics, Neuro, and General Surgery.
Integrates seamlessly into existing implant-based protocols and procurement pathways.
Minimizes clinical and financial fluctuations through a single, validated platform.
Reduces inventory redundancy by replacing fragmented, multi-vendor biologic portfolios.

STA™ is regulated under Section 361 of the Public Health Service Act as a structural tissue allograft intended for homologous use. Its classification has been reviewed by the FDA Tissue Reference Group (TRG), confirming alignment with minimal manipulation & homologous use. This level of regulatory clarity supports efficient Value Analysis Committee review and enables confident adoption across health systems. All products are sourced exclusively from AATB-accredited, FDA-registered tissue banks and processed under controlled conditions.
We are not a biologics company with multiple products. We are a structural tissue repair platform designed for health systems that delivers consistency across surgical, wound, and clinical environments.

STA™ is the anchor of our platform. A full-thickness, hydrated matrix designed specifically for the high-demand environment of the operating room.
Replaces multiple “boutique” surgical biologics with one validated structural standard.

Extending our core structural technology into the chronic and surgical wound care space. By maintaining the same architectural integrity, AyoVita™ provides the scaffold necessary for complex closure.
Streamlines the transition from the OR to outpatient wound clinics using a consistent biologic framework.

Purpose-built structural applications for environments where both structural performance and aesthetic refinement are critical.
Reduces the need for specialized aesthetic vendors by leveraging the existing health system platform.
Maintaining the full-thickness matrix that surgeons trust.
Protecting the signaling molecules inherent to the tissue.
Ensuring every implant performs with predictable excellence.
Clarity. Consistency. Confidence.
A new standard in tissue repair.